Your Questions, Answered
We’ve assembled the most frequently asked questions about Eversense into one place. Got a question that you can’t find the answer to? Contact us.
| Characteristics | Description |
|---|---|
| Class | II |
| Type | AC Input, 100-240Vac, 50/60Hx, 0.3-0.15A |
| DC Output | 5V DC, 1A (5.0 watts) |
| Moisture Protection (charging cradle) | IP22 (Protected from touch by fingers and objects greater than 12 millimeters. Protected from water spray or dripping water from vertical angle.) |
Removing an Eversense® CGM user from your list means you will no longer be able to remotely monitor their glucose data. To add a user back to your list, contact the Eversense® CGM user to send you a new invitation.
- Swipe the Eversense® CGM System user’s name on the Home Page to the left.
- Tap “Delete”. A warning will ask you to confirm you want to remove the Eversense® CGM user.
- Tap “Yes” to permanently remove this person from your list. The Home Page will no longer show the user in your list.
To add a user back to your list, contact the Eversense® CGM user to send you a new invitation.
Note: It can take up to 2 hours for the Eversense® CGM user to see that you are no longer in their Circle on their Eversense® CGM App.
The Eversense® App is a software application that runs on a mobile device (e.g., smartphone or tablet) and displays glucose data in a variety of ways. It also provides alerts based on the glucose settings you choose.
The Eversense® App screens layout will vary based on your mobile device’s model and/or operating system.
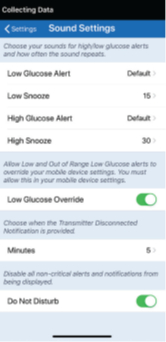
The Eversense® App has a “Do Not Disturb” option for non-critical alerts.
Tap Menu > Settings > Sound Settings to display the SOUND SETTINGS screen.
Activating the “Do Not Disturb” option will turn off all non-critical alerts and notifications, and these will not appear and the transmitter will not vibrate for these.
IMPORTANT TO NOTE: Critical alerts cannot be turned off.
Critical alerts include:
- Low Glucose
- Out of range low glucose
- Out of range high glucose
- Battery empty
- Sensor replacement
- High Smart transmitter temperature
- Low sensor temperature
- High sensor temperature
- Smart transmitter error
- Charge Smart Transmitter
- No Sensor Detected
- Transmitter Error
- Vibration Motor
- Transmitter Replacement (Day 396, 395, 366)
- Battery Error
- Sensor File Error
- Incompatible Transmitter
- Data Unavailable
All other alerts and notifications will be suppressed when Do Not Disturb is turned on.
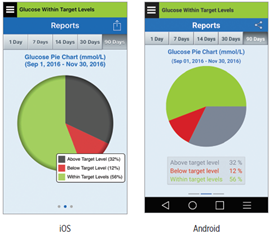
The Glucose Pie Chart report shows in graphical format what percent of your readings within a given time period are within, below or above your Glucose Target levels. You can choose the last 1, 7, 14, 30 or 90 days.
An internet connection is required to download the app and to receive remote glucose values for display on your Eversense® NOW App.
Still have questions?
The Eversense team is dedicated and ready to provide the answers and support you need. Simply click the link below.
The Eversense® E3 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to 180 days in persons with diabetes age 18 and older. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are still required for calibration primarily one time a day after day 21, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense E3 CGM System is a prescription device; patients should talk to their health care provider to learn more.
The Eversense® 365 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to one year in people (18 years and older) with diabetes. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are required for calibration one time a week after day 13, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense 365 CGM System is a prescription device; patients should talk to their health care provider to learn more.
For important safety information, see bit.ly/eversensesafety
Eversense, Eversense E3 Continuous Glucose Monitoring, Eversense 365 Continuous Glucose Monitoring, and the Eversense logo are trademarks of Senseonics, Incorporated. All other trademarks are properties of their respective owners and are used solely for informative purposes. No relationship or endorsement should be inferred or implied.
Apple Watch® is a product of Apple, Inc., and may be separately purchased from an authorized Apple retailer. Apple Watch is not included with the Eversense CGM System. Android is a trademark of Google LLC.
MKT-001692 Rev 1